Our collaborator Dr. Aboulwafa along with Dr. Zhang and Dr. Saier published a paper in the journal Microbial Physiology where .they studied protein-protein interactions of membrane proteins and their influence in sugar uptake systems. The title is: “Protein-Protein Interactions in the Cytoplasmic Membrane of Escherichia coli: Influence of the Overexpression of Diverse Transporter-Encoding Genes on the Activities of PTS Sugar Uptake Systems“. For your convenience, here is the link to PubMed.
Abstract
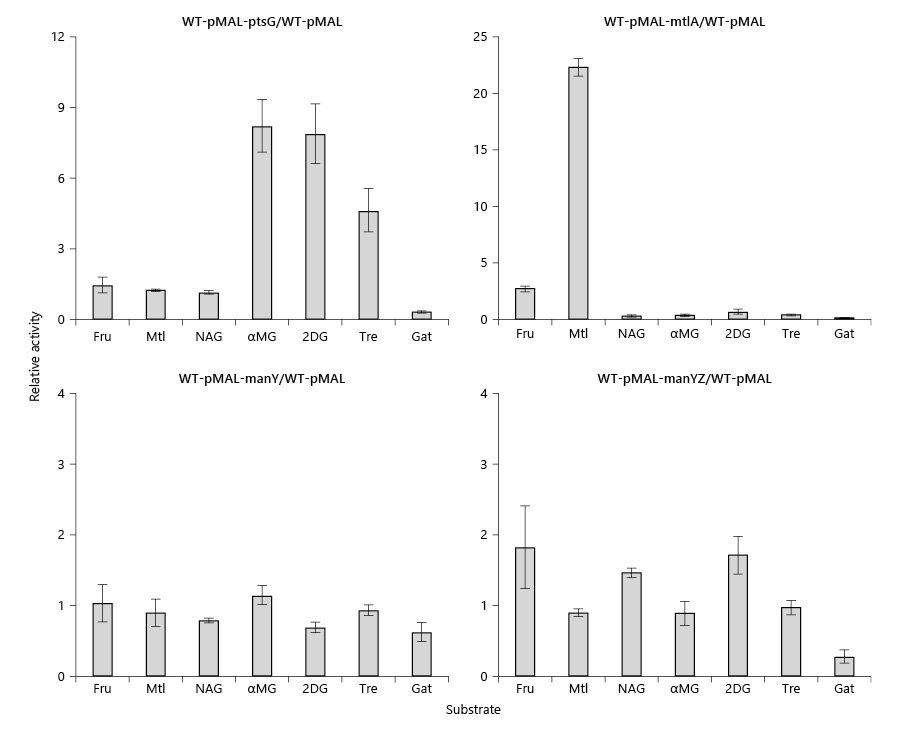
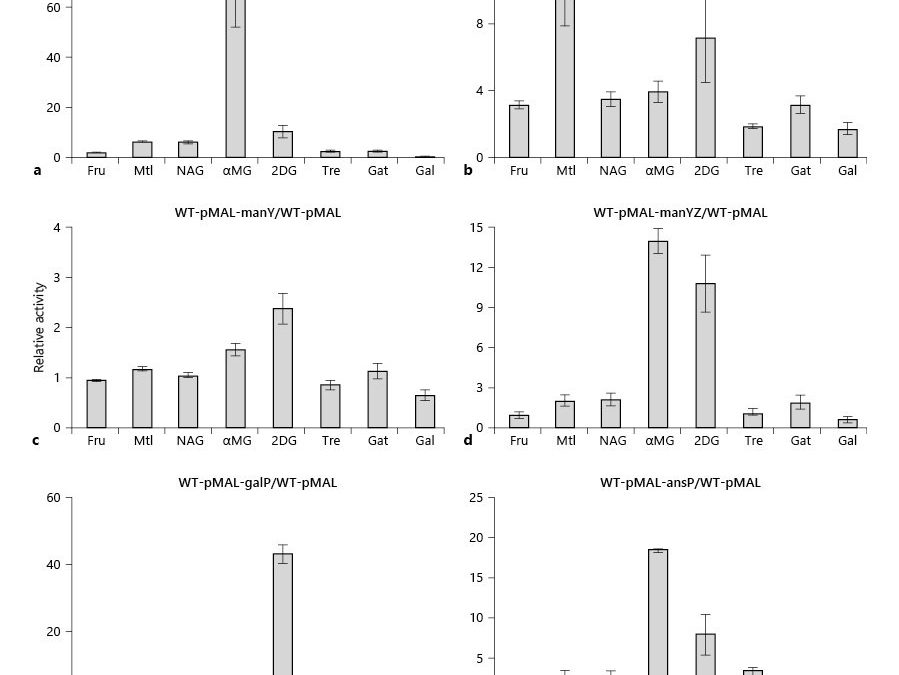
The prokaryotic phosphoenolpyruvate (PEP):sugar phosphotransferase system (PTS) concomitantly transports and phosphorylates its substrate sugars. In a recent publication, we provided evidence that protein-protein interactions of the fructose-specific integral membrane transporter (FruAB) with other PTS sugar group translocators regulate the activities of the latter systems in vivo and sometimes in vitro. In this communication, we examine the consequences of the overexpression of several different transport systems on the activities of selected PTS and non-PTS permeases. We report that high levels of these transport systems enhance the in vivo activities of several other systems in a fairly specific fashion. Thus, (1) overexpression of ptsG (glucose porter) selectively enhanced mannitol, N-acetylglucosamine, and 2-deoxyglucose (2DG) uptake rates; (2) overexpression of mtlA (mannitol porter) promoted methyl α-glucoside (αMG) and 2DG uptake; (3) manYZ (but not manY alone) (mannose porter) overexpression enhanced αMG uptake; (4) galP (galactose porter) overexpression enhanced mannitol and αMG uptake; and (5) ansP (asparagine porter) overexpression preferentially enhanced αMG and 2DG uptake, all presumably as a result of direct protein-protein interactions. Thus, it appears that high level production of several integral membrane permeases enhances sugar uptake rates, with the PtsG and ManXYZ systems being most consistently stimulated, but the MtlA and NagE systems being more selectively stimulated and to a lesser extent. Neither enhanced expression nor in vitro PEP-dependent phosphorylation activities of the target PTS systems were appreciably affected. The results are consistent with the suggestion that integral membrane transport proteins form an interacting network in vivo with physiological consequences, dependent on specific transporters and their concentrations in the membrane.